InnoScan 710IR microarray scanner: new publication
InnoScan 710-IR Facilitates Precision ADC Therapy in Breast Cancer
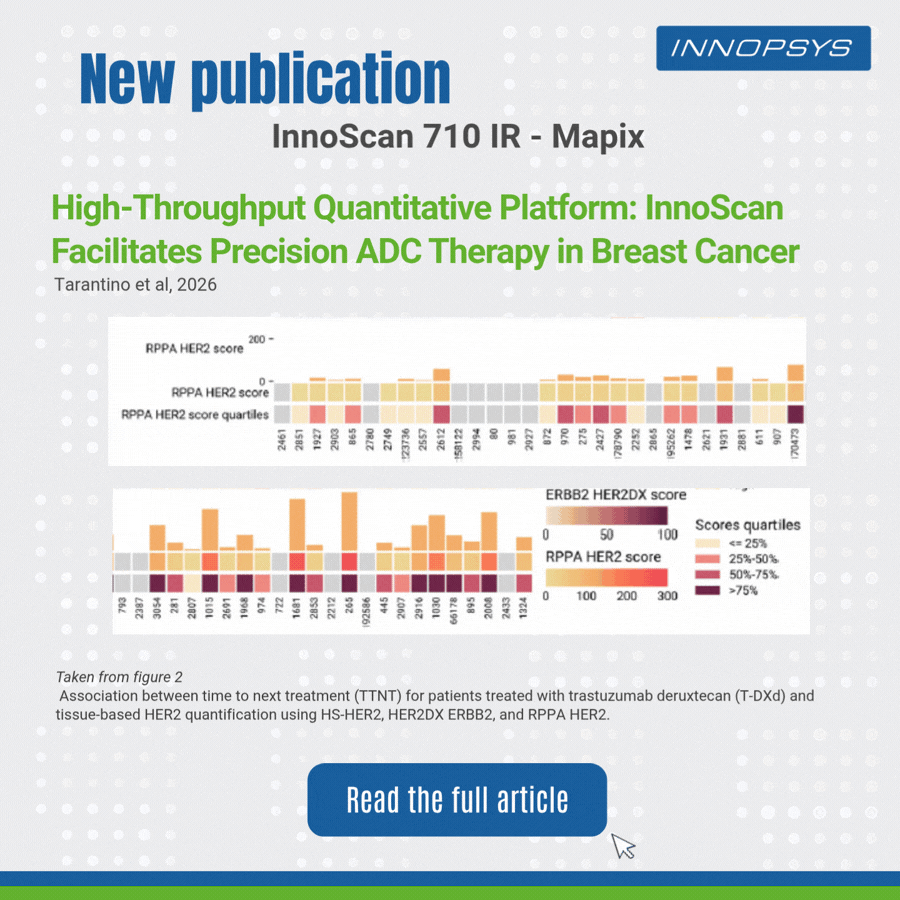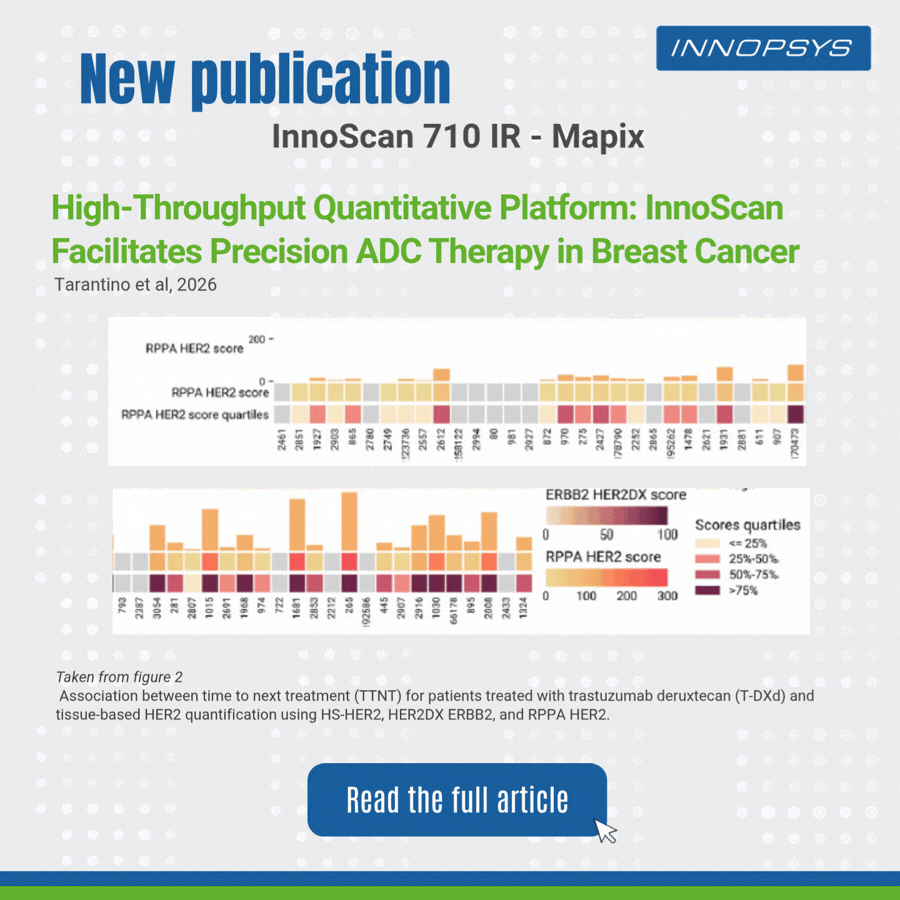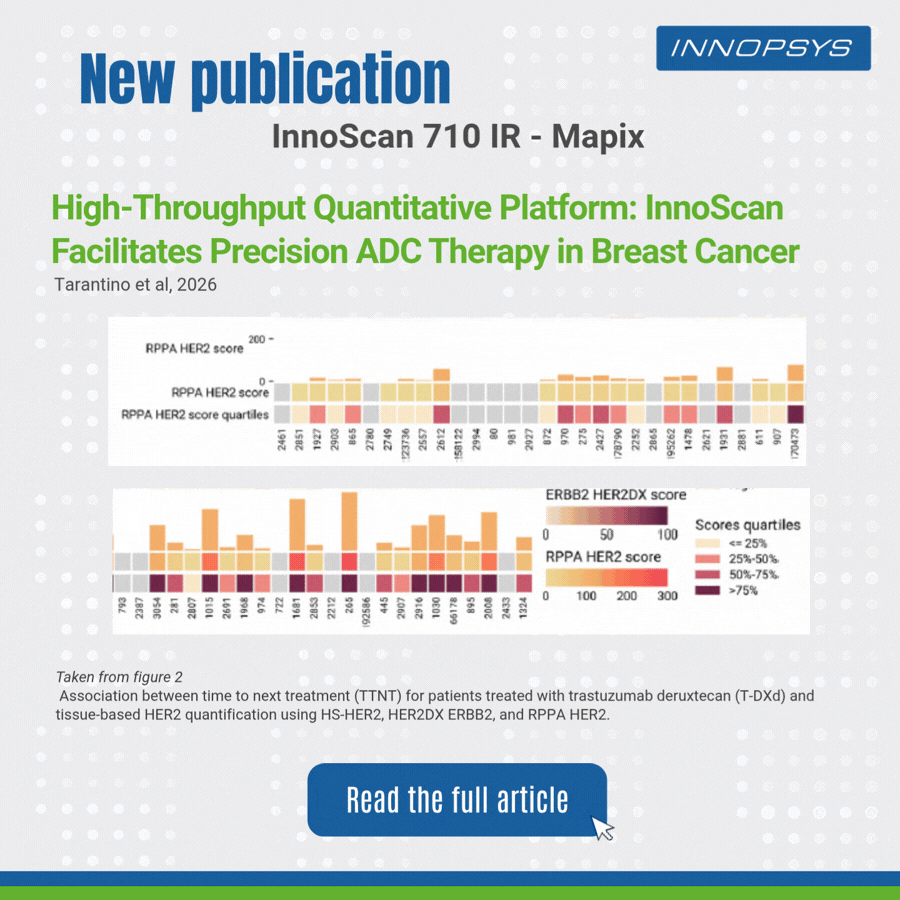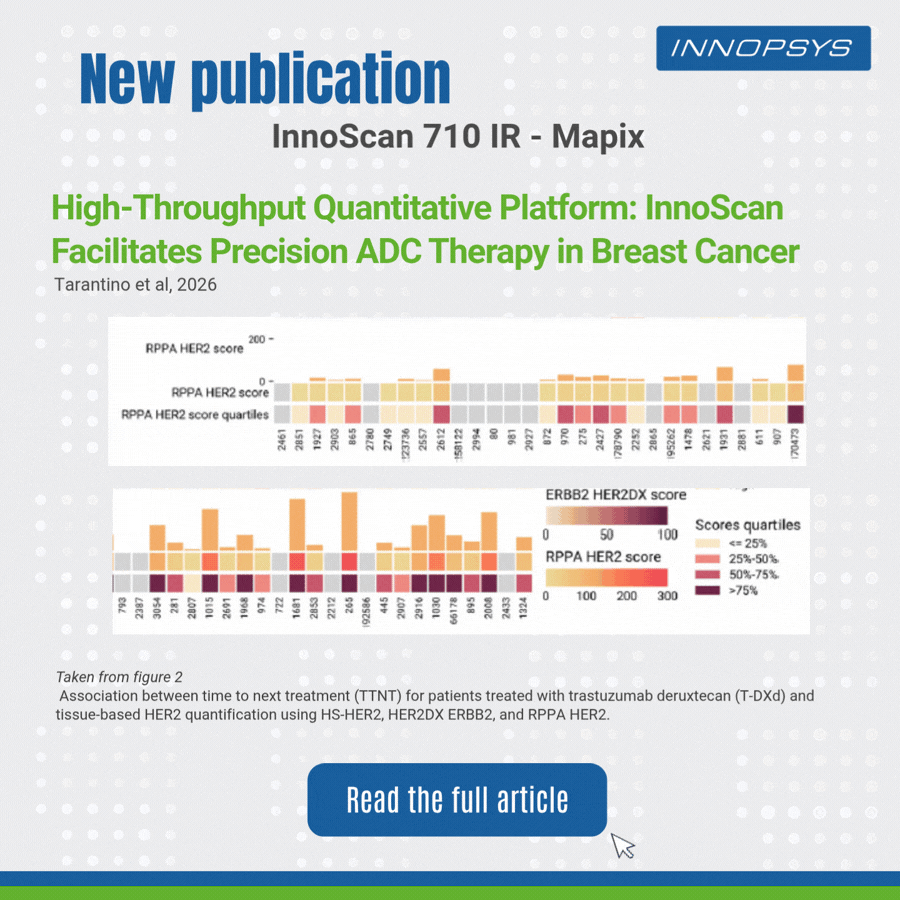
A clinical translational study published in npj Precision Oncology successfully developed a multi-platform HER2 quantitative scheme to accurately predict clinical benefits of T-DXd in breast cancer patients. The team adopted RPPA, HER2DX, DNADX and other technologies to assess HER2 status comprehensively. InnoScan 710IR (coupled with MAPIX analysis software)was responsible for digital signal reading of RPPA microarray, rapidly completing quantitative analysis of hundreds of sample spots and outputting standardized data for efficacy biomarker analysis. Results showed HER2 quantitative levels were significantly correlated with patient survival, and high TOPO1 expression indicated poor efficacy in HER2-negative patients. This study provides a translatable precise strategy for individualized ADC administration.
Original publication : Quantitative HER2 tissue and plasma profiling predicts the activity of trastuzumab deruxtecan for breast cancer | npj Precision Oncology
Keywords:
- HER2 quantification
- T-DXd efficacy prediction
- Reverse Phase Protein Array (RPPA)
- InnoScan 710IR
- Breast cancer precision medicine
TO Go Further with the HER2 analysis: think about InnoQuant
Did you know that our InnoQuant slide scanner can directly support high-sensitivity HER2 (HS-HER2) quantitative tissue analysis as described in this work from Tarantino et al.?
In this study, the HS-HER2 test relies on quantitative immunofluorescence to measure HER2 expression on a continuous scale (amol/mm²), demonstrating a strong association with clinical outcomes. This requires highly accurate and reproducible image acquisition, particularly because HER2 signal intensity spans a wide dynamic range across patient samples (Fig. 3). The use of tyramide signal amplification (TSA) further increases signal intensity, making it essential to avoid saturation while still detecting low-expression regions. In this context, a high dynamic range (16-bit) combined with low background noise -one of InnoQuant’s key strengths- is critical to ensure reliable quantification of both weak and strong signals within the same tissue.
In addition, the HS-HER2 workflow integrates multiplex immunofluorescence (e.g., DAPI, cytokeratin, HER2), enabling tumor segmentation and region-of-interest (ROI) analysis by pathologists. InnoQuant supports simultaneous multi-channel acquisition with minimal crosstalk, allowing efficient multiplexing without increasing acquisition time. Its ability to generate a full-resolution whole-slide image in a single pass enables seamless downstream analysis in digital pathology tools such as QuPath, which is consistent with the analysis pipeline used in the study.
It is important to note that quantitative assays such as HS-HER2 require high spatial uniformity and excellent reproducibility across the entire slide, particularly when comparing samples or converting fluorescence intensity to absolute protein concentration using calibrated standards. Thanks to its unique rapid linear scanning optical design, InnoQuant eliminates image stitching and achieves up to 98% illumination uniformity across the entire slide, reducing acquisition biases and eliminating the need for post-processing corrections.
Finally, the study highlights the need for efficient processing of multiple patient samples (e.g., a 51-patient cohort for HS-HER2 analysis). With its 24-slide capacity and fully automated workflow, InnoQuant enables high-throughput, reproducible batch acquisition with standardized protocols, making it well-suited for translational and clinical research applications
But the strongest asset of InnoQuant resides in its reproducibility and absolute quantitative results. We guarantee the stability of our lasers and our detectors (PMT) over time, for interslide and inter batch reproducibility. Our lifetime-guaranteed quality control slide, coupled with a fast Checkscanner click-button procedure, enables you to check the state of your scanner at any time. We are the only manufacturer to guarantee 100% quantitative fluorescent reproducible results.