Formed in 1999, Innopsys develops, manufactures and sells equipment and associated software for biotechnological & medical research laboratories combining cost-effectiveness with performance.
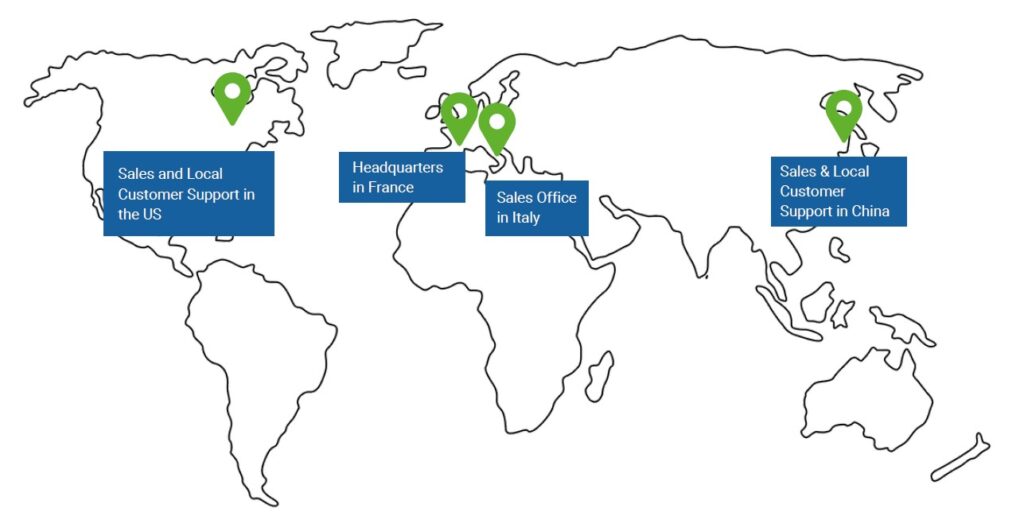
Innopsys sells its products in more than 50 countries through its worldwide distribution network and its United States subsidiary in Chicago.
Innovation is the core of Innopsys’ strategy; annually investing 30% of its turnover in R&D. Its own efforts combined with numerous academic and industrial partnerships make Innopsys a company with great potential in the market for the biotech products of tomorrow.
Products are entirely designed and manufactured on site in Carbonne, situated in the Midi-Pyrénées area of France according to ISO 13485 requirements. The Innopsys team is comprised of mostly scientists and engineers and is distinguished by its cutting-edge skills in a variety of scientific and technical fields including optics, micromechanics, onboard electronics, computing, nanotechnology and biology.
Life Science

Since 2006, Innopsys has offered a complete range of InnoScan microarray scanners, and became a leader in its market. In late 2023, Innopsys launched InnoQuant, an innovative tile-free fluorescence scanner designed for tissue multiplexing, bringing the advantages of line scanning to the fields of histotechnology and immunocytochemistry.

Our Commitments
Since 2009, Innopsys has been certified ISO 13485 (Medical Devices) for its quality management system.
ISO 13485 is intended for organizations involved in the design, production, installation and maintenance of medical devices and related services.
Manufacturing Process
Control and rigor
By controlling all stages for the product (from design to full-scale production), Innopsys responds to all its customers’ needs.
In our manufacturing plant, Innopsys teams carry out tests, assembly and settings for each component of its equipment.
Our production is based on rigorous traceability. It includes identifying every component, sub-assembly and manufactured machine part as well as recording inspection and setting information.
Manufacturing is computerized and is easily monitored and accessible in an Innopsys Intranet database.
At the end of manufacturing, equipment is validated using two types of tests:
- stability tests for optical settings across a range of temperatures over 8 hours in an oven
- reliability tests during operation in an endurance cycle lasting 5 days

Customer Support
A team that listens
Members of Innopsys Customer Support are available, before and after a sale, to answer questions pertaining to the following:
- commercial
- technical
- logistics
The support team’s responsiveness and desire to meet and exceed customer expectations ensure that it can responds to specific and individual projects swiftly.
After a sale, installation, training and support are provided for our products on our customers’ premises by Innopsys and/or our export partners.
Organisation of Customer Support
Customer Support is organised between our partners and our team. Its aim is to offer a high quality support service, anywhere in the world and for all Innopsys products.
Your local contact will provide a rapid response to all your commercial and logistical questions, as well as all your technical questions.
Our scanner and software has been designed to collect data remotely to facilitate and speed-up a remote diagnostic.
Ethic and corporate social responsibility
Since its inception, Innopsys has pursued a responsible approach to the environment. Our premises in Carbonne, France were constructed in 2004 using a High Environmental Quality (HEQ) approach. The building was designed in accordance with bio-climatic principles (sunshine exposure control), using non-polluting, low-energy-cost materials (wood, gas heating, bio-friendly paint and natural insulation).
Our Commitment to Sustainability Recognized with EcoVadis Silver Rating.
In December 2024, we were proud to be awarded the EcoVadis Silver Medal, placing us among the top-performing companies in terms of corporate social responsibility (CSR). This independent recognition reflects our ongoing commitment to ethical business practices, environmental stewardship, and responsible supply chain management. For our clients, this rating offers a clear guarantee: by working with us, you are partnering with a company that meets rigorous international standards for sustainability and transparency. It reinforces trust, reduces risk, and supports your own sustainability goals, all while contributing to a more responsible and future-focused value chain.

Innovations & Partnerships
Innovation is at the heart of Innopsys way of being. Not only in its cutting-edge technologies and products but also in its management and sustainable development.
Pioneer in its regional area with an ecological building made of wood, Innopsys offers a pleasant working environment as well. The consistent team shares its free time to play sport or participate to the company’s garden.
Each year since it was created in 1999, Innopsys has invested more than 30% of its turnover in Research & Development.
Today, 60% of its employees are scientists and engineers specialised in their fields: optics, micromechanics, servo mechanisms, onboard electronics, software, biology or nano-technologies.
But these specific skills are also complemented by the expertise of its joint lab – Biosoft – with LAAS-CNRS. Biosoft is a 50m² open lab devoted to welcome collaborations by giving a free access to most advanced Innopsys technologies. It is also where the most innovative works take place, benefiting from CNRS facilities: class 100 clean room, cell culture room, 3D-Lithography and photolithography equipment and characterization equipment (AFM, quartz microbalance…).
Currently the main topic of study in Biosoft is the single-cell analysis. It is performed thanks to the InnoScan 1100 AL for analysis, the microfluidic devices developed by Biosoft and the combined expertise of both teams.
Innopsys participated to 7 collaborative projects in 5 years, working with 15 different partners. The addressed topics are mainly focused on bio-sensors for IVD market, environment and life sciences.
Innopsys holds numerous international patents which are exploited in its products and processes.
